About Hypogonadism and TRT
Balanced Hormones, Better HealthRestoring T levels may improve his overall health
Hypogonadism affects about 30% of American men 20 years of age and older and increases in prevalence as testosterone (T) levels decline with age.1,2
Symptoms of Low Testosterone LevelsRecognize Low-T 3,4
Studies show that restoring T levels may improve signs and symptoms associated with testosterone deficiency.3,9,a
Chronic disease and comorbidities are significantly more prevalent among men with T deficiency, including obesity, type 2 diabetes, cardiovascular disease, liver disease, and depression.1,3,5-8
aA series of observational studies evaluating the effect of testosterone therapy on various health conditions.11,12,15
Low Testosterone Therapy
Testosterone Replacement Therapy (TRT)Restoring the balance
Testosterone replacement therapy (TRT) to get T levels back to a normal range About 300 to 1000 ng/dL is considered a normal testosterone level (< 300 ng/dL is considered testosterone deficiency). The goal or TRT is to keep T levels within the normal range, avoiding serum concentrations that may be too low to support efficacy or too high, thereby increasing the risk of adverse effects.1,3,16 usually consists of testosterone patches, gels, pellets, or injections and, for some, oral medications.9
Characteristics of different TRTsWhich TRT is appropriate for your patients?
| KYZATREX™ | Injection | Implanted Pellets | Gel | Patches | |
|---|---|---|---|---|---|
| Oral / Easy to swallow | |||||
| Invisible | |||||
| No mess | |||||
| Painless | |||||
| Flexible Dosing |
KYZATREX™ is a novel testosterone undecanoate that has a smooth pharmacokinetic profile, strong safety profile, and wide dosing range for individualized TRT.
Daily oral dosing
Taking KYZATREX™ is easyNo gel, injection, pellet or patch, Just two oral daily doses, down the hatch
Just two oral doses of KYZATREX™ a day with food a can restore testosterone to normal levels and keep them there.
FlexibleDifferent doses for different dudes
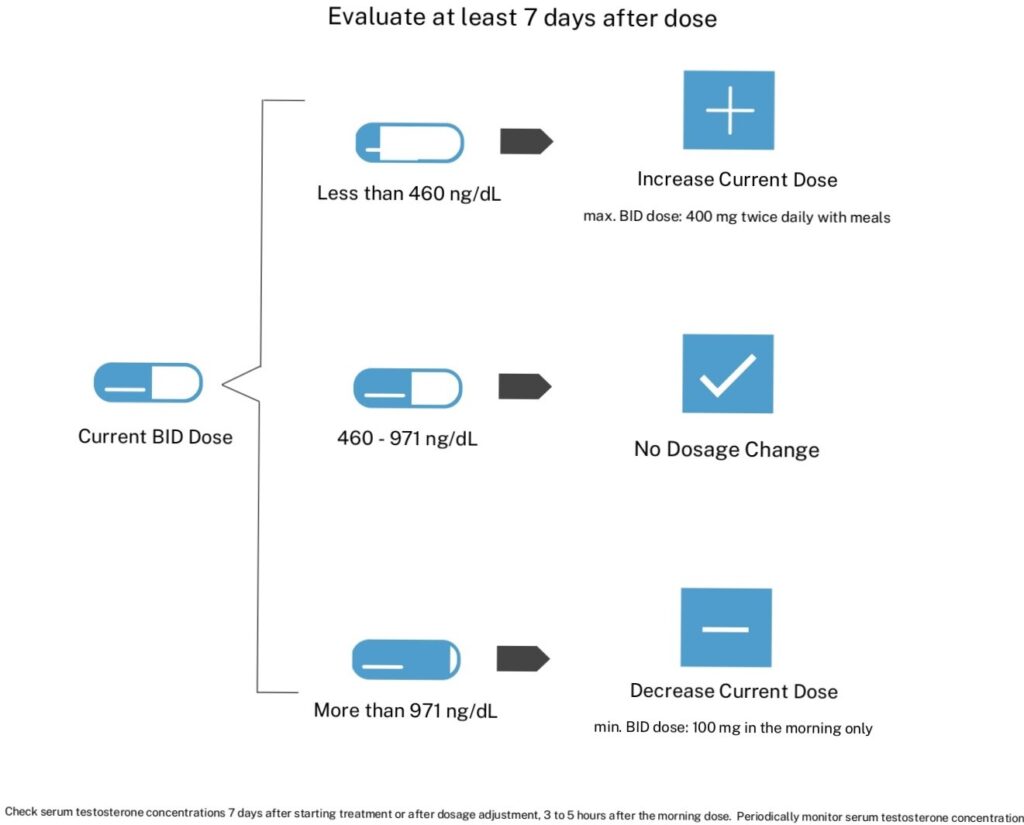
Meet your patients testosterone needs with a range of KYZATREX™ doses. The recommended starting dose of KYZATREX™ is 200 mg orally BID with the morning and evening meals. After 7 days of starting treatment, dose may be adjusted to meet target testosterone concentration.
And BioavailableFormulated to drive absorption to improve clinical results
KYZATREX™ is formulated to utilize lymphatic absorption to bypass first-pass metabolism and drive uptake. With the use of phytosterols as GRAS excipients to drive absorption, KYZATREX™ was designed for clinical efficacy and safety.
a KYZATREX™ must be taken with food.
Titration Guide
Lymphatic Absorption KYZATREX™ is well absorbed from the GI tract when taken with food.
About KYZATREX™
KYZATREX™, put to the test – The power of balance
96%Effective!
In a clinical study of 139 men with low testosterone, 96% had normal testosterone levels at Day 90.*
*End of clinical study completers
Most common adverse effects in clinical studies a
Side effects in ≥2% of Patients Receiving KYZATREX™ (n=155)
- Hypertension 2.6%
In a 12-month, open-label clinical study, men who received KYZATREX™ 200 mg QD to 400 mg BID (n = 202) reported the following additional adverse reactions: headache, arthralgia diarrhea, hemoglobin increased, anxiety, constipation, peripheral edema, and PSA increased.
PSA, prostate-specific antigen.
a In the 6-month extension study of 155 patients taking KYZATREX™, 1 patient discontinued because of an adverse reaction (acne).
It’s all in the delivery
Each KYZATREX™ oral capsule is uniquely formulated KYRATREX is the first prescription medicine to use phytosterols (naturally occurring compounds found in plants) to boost absorption rates. Phytosterols are generally recognized as safe (GRAS) excipients. to be easily absorbed and bypass your liver. That means it works efficiently, without causing liver damage.
Blood Pressure
Side effects in ≥2% of Patients Receiving KYZATREX™ (n=155)
- High Blood Pressure 2.6%
In a study of 155 patients taking KYZATREX™ for six months, only one discontinued because of a side effect, in this case, acne.
-
Endpoint
MRS-TU-2019EXT
Study (N = 155) - ABPM17
-
Change in 24-hour mean SBP from baseline after 4 months of KYZATREX™, mmHg (95% CI)
1.7
(0.3-3.1) -
Change in 24-hour mean SBP from baseline after 6 months of KYZATREX™, mmHg (95% CI)
1.8
(0.3-3.2) - In-clinic Blood Pressure Cuff
-
Change from baseline in mean SBP after 4 months of KYZATREX™, mmHg (95% CI)
2.7
(0.9-4.5) -
Change from baseline in mean SBP after 6 months of KYZATREX™, mmHg (95% CI)
2.4
(0.6-4.2) - ABPM, ambulatory blood pressure monitoring; CI, confidence interval; SBP, systolic blood pressure.
- Diastolic blood pressure was not significantly different from baseline.
- A total of 5/155 (3.2%) participants started taking new antihypertensive medication after study start. No participants had a dose increase in their antihypertensive medication by the end of treatment.
- A history of antihypertensive treatment and diabetes mellitus at baseline were significant factors related to ABPM SBP increases.
Get KYZATREX™
Submit eRx on EMR for Fulfillment through Eagle Specialty Pharmacy: (Only available through Eagle Specialty Pharmacy)
- NPI: 1487905840
NCPDP: 5711975
Name: Eagle Pharmacy
Address: Lakeland, FL 33810
Instructions for Patient to Order Online
- Enroll at www.kyzatrexdirect.com
OR - Give KYZATREX™ Direct Card to patient


-
Price: $159/month – Cash Only (Not covered by insurance)
For office collateral, including KYZATREX™ Direct Cards, please email info@mariuspharma.com
Call 888-994-1950 for Assistance
Support & Resources
Materials Coming Soon


